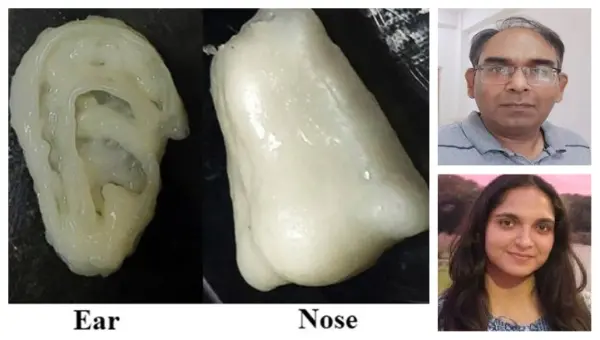
Rourkela: Researchers at the National Institute of Technology (NIT), Rourkela, have created and patented a new bio-ink that could significantly advance 3D bioprinting for tissue engineering, particularly in repairing bone and cartilage.
The innovative material addresses a major limitation in current bio-inks by successfully combining high mechanical strength, excellent biological compatibility, and superior printability — properties that have traditionally been difficult to achieve together.
Led by Associate Professor in the Department of Biotechnology and Medical Engineering Devendra Verma, the team, including research scholars Shreya Chrungoo and Dr Tanmay Bharadwaj, developed a high shape-fidelity composite bio-ink made from Bovine Serum Albumin (BSA), Sodium Alginate, and polyelectrolyte complexes of gelatin and chitosan (PEC-GC) that can be effectively used in bone and cartilage repairing. This protein-polysaccharide blend creates a bioactive system that supports cell growth while maintaining structural fidelity during and after the printing process.
The research has b
een published in the International Journal of Biological Macromolecules, and the team has secured an Indian patent titled ‘A High Shape-Fidelity Protein-Polysaccharide Composite Bioink for 3D Bioprinting’.
“Our goal was to bridge the long-standing gap between printability and biological performance in bio-inks. By integrating protein–polysaccharide interactions with nanofibrous complexes, we have developed a system that not only prints with high precision but also actively supports cellular functions and tissue regeneration. This brings us a step closer to clinically relevant bioprinted constructs,” Prof Verma said.
Laboratory tests showed promising results with the developed bio-ink mimicking the extracellular matrix of bone tissue, providing sites for cell attachment, and promoting cell adhesion, proliferation, and overall biological response. Scaffolds containing 2% PEC-GC achieved over 90% cell viability, demonstrated strong mechanical properties for shape retention, and promoted bone tissue formation and collagen synthesis.
Shreya Chrungoo, the research scholar, emphasised the practical potential: “The developed bio-ink offers a versatile platform for fabricating patient-specific scaffolds with precise geometry and biological functionality. Its ability to support high cell viability and tissue-like behavior makes it promising for applications in regenerative medicine.”
The team now plans to undertake animal studies to evaluate safety and efficacy of the developed bio-ink, followed by clinical studies for validation.
If successful, the technology could open new doors for personalised regenerative treatments, enabling the creation of customised tissue scaffolds tailored to individual patients.